Managing ethics in H2020 projects

It is nowadays a fact that in all EU Horizon projects, ethics is an inherent aspect of all research activities while ethics compliance is considered critical to achieving research excellence. A process has been established for all activities funded under H2020 aiming to assess and address the ethical dimension starting from the proposal stage of a project and continuing until the end of the project duration. The process is known as Ethics Appraisal Procedure. It starts at the proposal stage with the Ethics Self-Assessment and aims to ensure that the provisions on ethics in the H2020 regulation and in the Rules for Participation are respected.
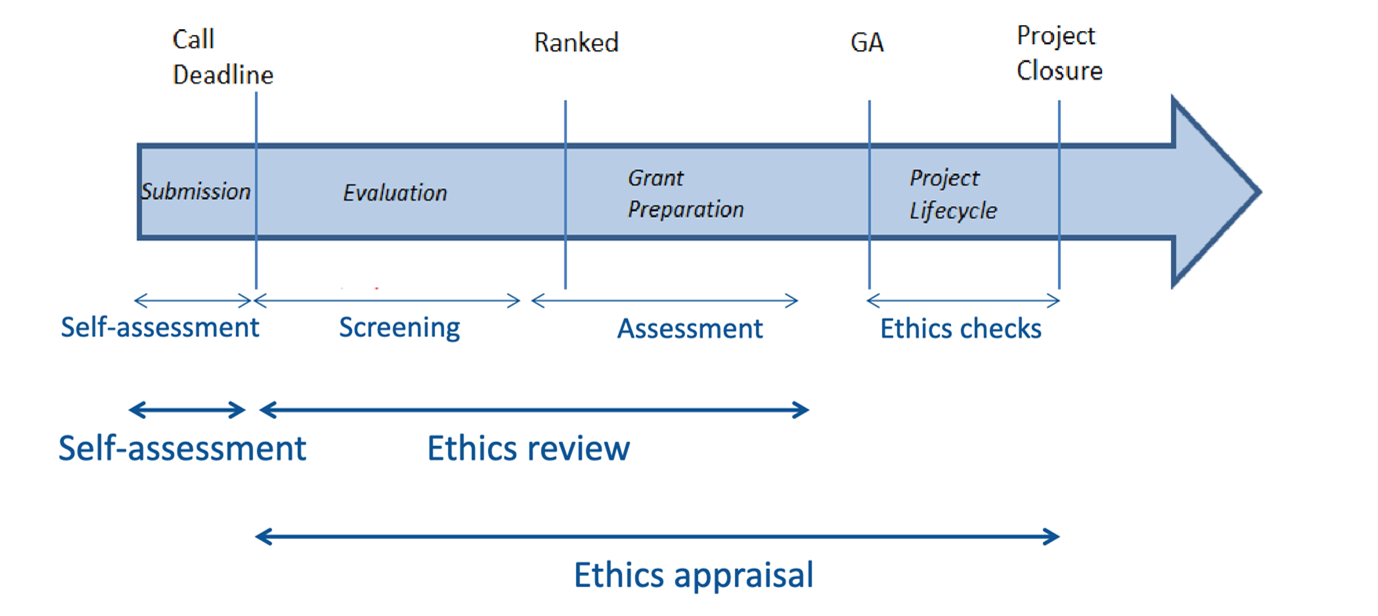
MARVEL is approaching the end of the 1st year, a year full of activities on ethics (deliverables, meetings, discussions, etc.). After the successful scientific evaluation of the proposal that led to the decision of the EC to fund MARVEL, the Ethics Review Procedure was carried out. As a result, the project received a Conditional Ethics Clearance and ethics requirements: 10 scheduled for fulfillment during the grant agreement preparation and another 10 during the MARVEL active phase. These requirements are related to data processing, data misuse, risk of mass surveillance, anonymisation/pseudonymisation, secondary use of data, research activities outside EU, informed consent, experimental subjects, and algorithmic bias and trustworthy AI. During the GA preparation phase, the MARVEL consortium successfully addressed the pre-grant ethics requirements. The post-grant ethics requirements were grouped in six deliverables under WP9. Their submission started in M2 and will be concluded by the end of the first year (M12).
How we manage ethics in MARVEL
So, how do we intend to guarantee ethics, legal, and privacy compliance while achieving high-quality results in MARVEL? Below, we summarise a list with tips and recommendations on how we manage ethics in MARVEL, aiming to share the experience we have gained up to now with other EU Horizon projects.
- Assign a European Privacy & Data Protection Law expert to serve as the Data Protection Manager (DPM) of the project: It is very important while a proposal is being formalised to include in all the preliminary discussions a legal partner, who will provide advice and guidance on all the ethical aspects that the proposal touches upon. If your project deals with many ethical, legal, and privacy issues, dedicate a specific task to ensure compliance that will address potential ethics requirements, collaborate with WP and Task leaders, provide supervision and recommendations, and liaise with partners’ data protection officers (DPOs) throughout the entire project life cycle. For MARVEL, this role is assigned to Privanova, a consultancy created by former INTERPOL professionals focusing specifically on outstanding ethics and GDPR compliance management.
- It is not only the responsibility of the Project Coordinator and the DPM to meet project’s compliance goals. All beneficiaries should be aware of the ethical risks and actively assist. The PC and the DPM should request input from all related partners when drafting the ethics deliverables. Partners should also understand the meaning of the ethics requirements. Besides translating the ethics requirements for other partners so that they can understand and act on them, it is often useful to provide them legal documentation and official guidance documents on which the Ethics Appraisal Process is based.
- Dedicated meetings on privacy, ethics, and legal issues should be organised on a regular basis. Set up monthly meetings to review and monitor issues related to ethics. Ethics compliance should be seen as an integral part of the overall project management. It should also result in adequate feedback on potential ethics challenges and, because of this must be connected to the risk management component of the project. All partners are equally responsible to meet ethical and legal requirements for the work they undertake in the project.
- External Ethics Advisory Board: Independent oversight is vital to ensuring ethics, legal, and privacy compliance: Invite independent experts, external to the consortium with relevant background, including Privacy & Data Protection Law experts. The EAB will oversee, advise, assess and, when applicable, raise concerns to the Project Coordinator and consortium partners on relevant ethical issues within the project. Frequent interaction must be achieved by inviting the EAB members to attend project meetings and review deliverables where they can offer their comments and feedback. In-person meetings should be scheduled at least annually giving the EAB members the opportunity to help the consortium prepare for the project review.
- Complying with ethics requirements is a continuous process that starts even before the project enters the active phase and continues until the last day, so monitor your project activities constantly and stay up to date on possible new EU releases on the topic.
These are the tips we have collected from the first year of the project. However, as already mentioned, ethics compliance is a continuous process and of great importance. In the context of MARVEL, many activities are planned for the following years to ensure compliance, so stay tuned and read our news! More recommendations and lessons learned will follow in the coming months!
Menu
- Home
- About
- Experimentation
- Knowledge Hub
- ContactResults
- News & Events
- Contact
Funding

This project has received funding from the European Union’s Horizon 2020 Research and Innovation program under grant agreement No 957337. The website reflects only the view of the author(s) and the Commission is not responsible for any use that may be made of the information it contains.

